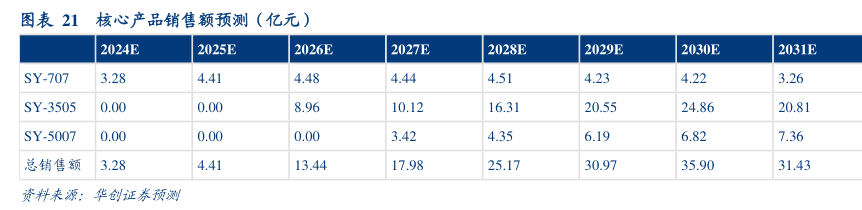
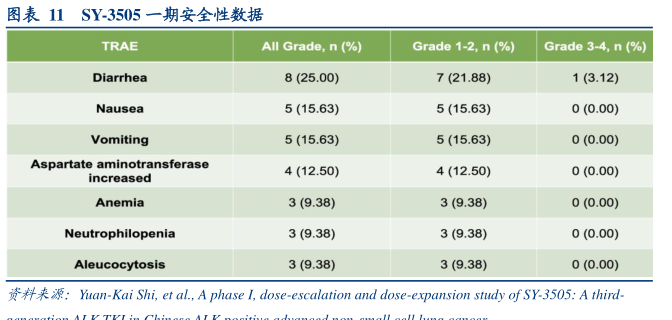
图表内容
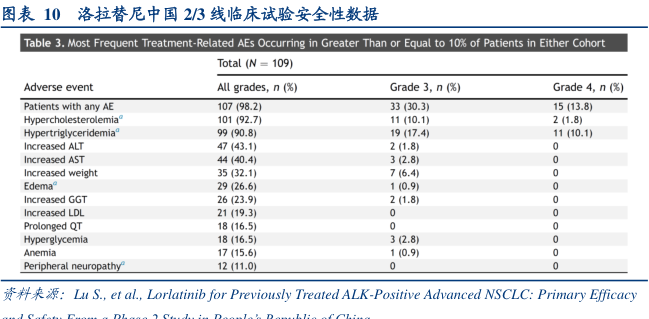
图表10洛拉替尼中国23线临床试验安全性数据
Table 3.Most Frequent Treatment-Related AEs Occurring in Greater Than or Equal to 10%of Patients in Either Cohort
Total (N 109)
Adverse event
All grades
n (%
Grade 3
n (%
Grade 4
n (%
Patients with any AE
107(98.2)
33(30.3
15(13.8)
Hypercholesterolemia
101(92.7)
11(10.1)
2(1.8)
Hypertriglyceridemia
99(90.8)
19(17.4)
11(10.1)
Increased ALT
47(43.1)
2(1.8)
Increased AST
44(40.4)
3(2.8)
35(32.1)
7(6.4)
Edema
29(26.6)
Increased GGT
26(23.9
2(1.8)
Increased LDL
21(19.3)
Prolonged QT
18(16.5)
Hyperglycemia
18(16.5)
3(2.8)
Anemia
17(15.6)
Peripheral neuropathy"
12(11.0)
资料来a源:LuS.
etal
Lorlatinib for Previously Treated ALK-Positive Advanced NSCLC:Primary Efficacy