图表内容
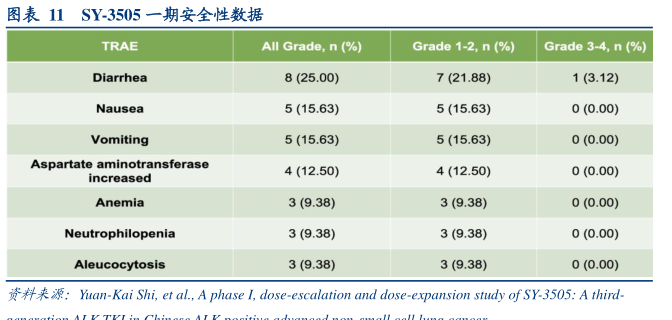
图表11SY-3505一期安全性数据
TRAE
All Grade
n(%)
Grade 1-2
n (%
Grade 3-4
n (%
Diarrhea
8(25.00)
7(21.88)
1(3.12)
Nausea
5(15.63)
5(15.63)
0(0.00)
Vomiting
5(15.63)
5(15.63)
0(0.00)
Aspartate aminotransferase
increased
4(12.50)
4(12.50)
0(0.00)
Anemia
3(9.38)
3(9.38)
0(0.00)
Neutrophilopenia
3(9.38)
3(9.38)
0(0.00)
Aleucocytosis
3(9.38)
3(9.38)
0(0.00)
資料来a源:hua-Kai Shi
etal.
A phase I
dose-escalation and dose-expansion study of SY-3505:A third-.