图表内容
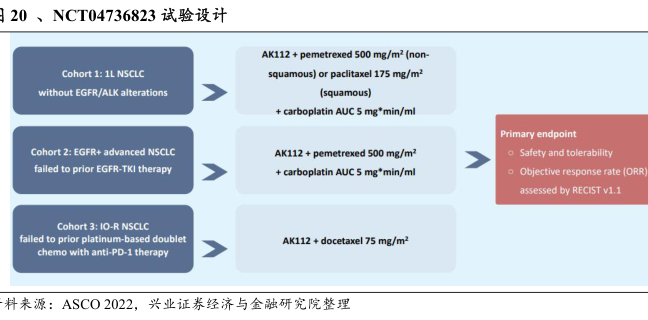
20、NCT04736823试验设计
AK112+pemetrexed 500 mg/m2 (non-
Cohort 1:1L NSCLC
squamous)or paclitaxel 175 mg/m2
without EGFR/ALK alterations
(squamous)
carboplatin AUC 5 mg*min/ml
Primary endpoint
Cohort 2:EGFR+advanced NSCLC
AK112+pemetrexed 500 mg/m2
o Safety and tolerability
failed to prior EGFR-TKI therapy
carboplatin AUC 5 mg*min/ml
o Objective response rate [ORR
assessed by RECIST v1.1
Cohort 3:1O-R NSCLC
failed to prior platinum-based doublet
AK112+docetaxel 75 mg/m2
chemo with anti-PD-1 therapy
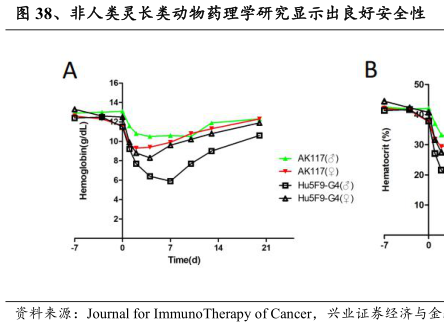
料来源:ASCO2022
兴业证券经济与金融研究院整理